Prediction of the Exit Site of Ventricular Tachycardia
Based on Different ECG Lead Systems.
Computing in
Cardiology 2017.
 Gary and
Bill Sanders Poster Award
Gary and
Bill Sanders Poster Award
links
abstract
Catheter ablation can permanently cure life-threating cardiac arrhythmia. Although the catheter position inside the heart can be precisely monitored, there is a lack of automated tools to guide the catheter tip to the arrhythmia exit site.
We studied the effectiveness of a computer-based method for exit-site localization based on a small number of ECG leads. We tested the influence of lead selection on localization accuracy.
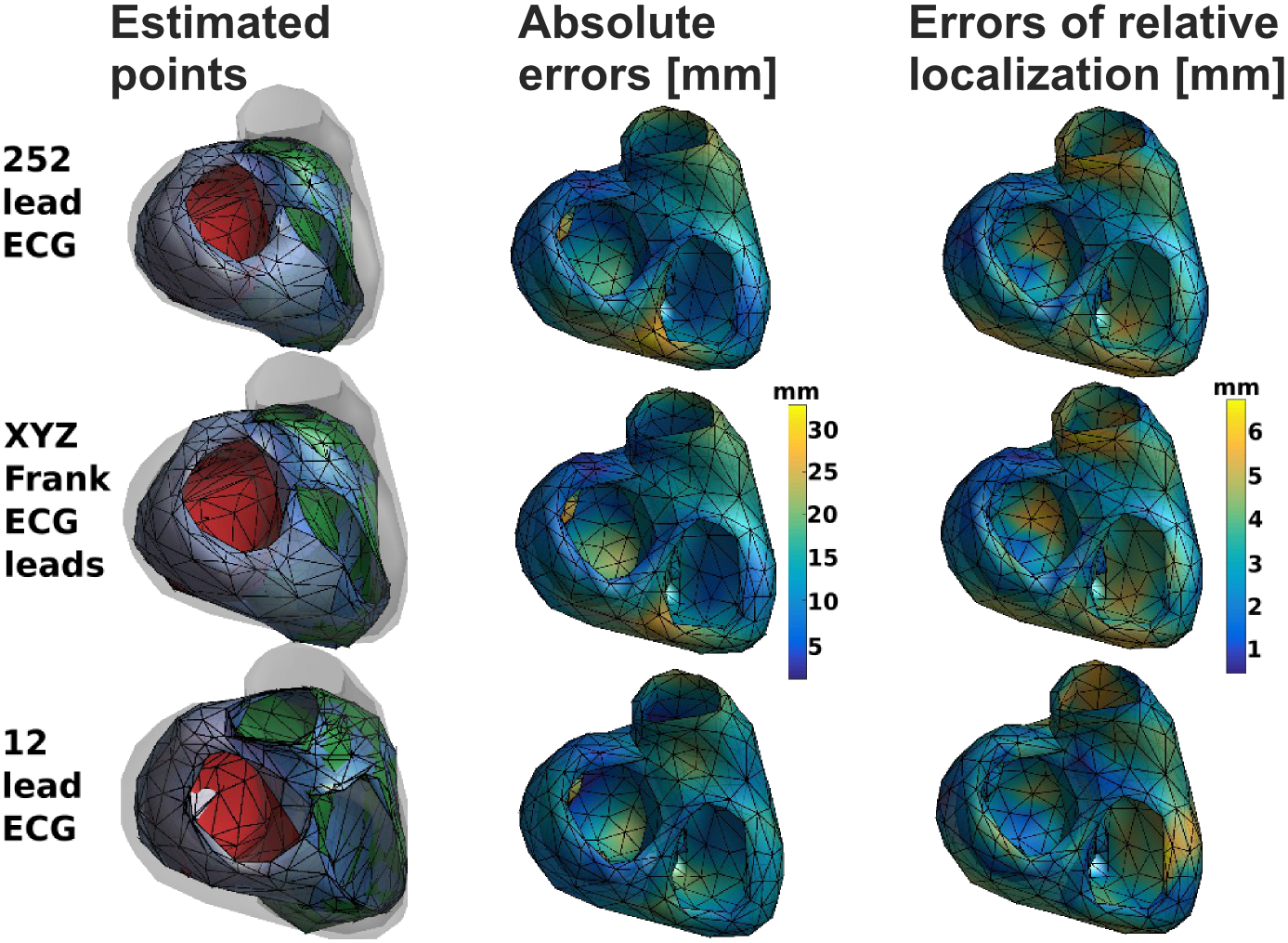
The method works on any set of 3 or more ECG leads. The QRS complex integral (QRSi) of an ectopic beat is reduced to principal components treated as coordinates of the exit site in ECG space and then projected to real space by a linear transformation. The accuracy of the method was tested on 5 patient-tailored models of human heart and torso. For each model ~500 simulations were run, each for different stimulus location. All locations were later estimated from simulated surface ECGs. The absolute and relative (to a neighboring stimulation site) localization errors (in mm) were computed for a 252-lead ECG, standard 12-lead ECG, and Frank VCG.
The group mean absolute and relative localization errors were: 11.5 (SD=2), 2.6 (SD=0.3) for 252-lead ECG; 12.4 (SD=1.8), 2.7 (SD=0.2) for 12-lead ECG; and 11.7 (SD=1.7), 2.7 (SD=0.2) for Frank VCG. The algorithm performed better for left ventricle than for right ventricle: for Frank VCG the group mean absolute errors were 10.9 (SD=1.7) and 12.4 (SD=1.7), respectively.
This study suggest that proposed method can predict exit sites with a precision in the order of a centimeter. Low values of relatives errors suggest opportunity for algorithm improvement. The use of vectorcardiographic leads is enough to obtain a precision comparable to full 252-lead ECG.
funding
The research leading to these results has received funding from the European Research Council under the European Union's Seventh Framework Programme (FP/2007-2013) / ERC Grant Agreement nr. ERC-2012- ADG_20120314.
This work was granted access to HPC resources of CINES and CEA-TGCC under GENCI allocation x2016037379.