Research Topics
cardiac resynchronization therapy
Heart-failure patients can be treated by "resynchronization therapy" with a biventricular pacemaker. It is important and challenging to predict which patients will benefit. The electrocardiogram plays an important role in the selection process. Unfortunately, several pertinent disease parameters affect the same electrocardiogram features. We are presently creating several highly detailed and physiologically accurate patient-tailored heart models with which we attempt to reproduce the electrocardiographic development of these patients during treatment. By combining electrocardiographic, imaging, and simulation data we attempt to improve understanding of the physiological changes in these patients and their effects on the electrocardiogram.
read more
diagnosis of atrial fibrillation substrate
Atrial fibrillation (AF) is a self-sustaining disease. Its progressive nature is presumably related to a slow but steady process of structural remodeling, which follows a relatively brief period of electrophysiological remodeling. The structural remodeling process is characterized by hypertrophy, activation of fibroblasts, enhanced collagen deposition, and changes in the distribution and function of gap junctions. These changes cause a heterogeneous deterioration of the atrial tissue's capacity to propagate the activation impulses. Therefore, the complexity of the propagating activation wavefront could be a measure of the disease stage, which plays an important role in the choice of an appropriate treatment. It would be desirable to infer this complexity from the electrocardiogram (ECG) measured at the body surface.
The Physiology department at Maastricht University tests and develops methods to assess the state of remodeling, based on the assumption that a more remodeled state leads to more chaotic activation, and thus to more chaotic ECG signals. I contribute to this research by performing AF simulations in a highly realistic model of the human atria in which the state of remodeling can be varied. The complexity of the atrial propagation and the ECG signals are quantified and related to each other to test the proposed methods.
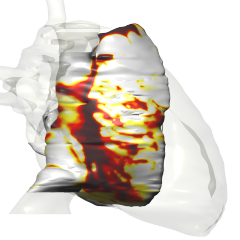
model development
Due to the complex anatomy and physiology of the heart, the relation between cardiac pathology and clinically measurable signals is often difficult to establish. I use large-scale mathematical modeling and quantitative signal analysis to explore this relation.
The main component of this development work is propag, a code for the simulation of cardiac electrophysiology. Originally designed to run on shared-memory systems with a moderate number of processors (32 to 64 at the time), it was initially parallellized with OpenMP. At the Institute of Computational Science in Lugano, the code has been upgraded to a "hybrid" MPI-OpenMP parallellization. The MPI parallellization allows it to run on distributed-memory computers with thousands of processors, while the OpenMP inner loops allow it to be more efficient on systems where many compute cores share their memory.

interaction of structural and ionic heart disease
Several cardiac arrhythmic syndromes have been attributed to mutations in the genes encoding cardiac ion channels. In many subjects, however, no mutations have been established yet. On the other hand, structural damage to the heart muscle (cardiomyopathy) has been observed in many of these subjects. The combination of structural and ionic abnormalities can disturb the propagation of the activating impulse, and can lead to arrhythmia. Research that I have done in collaboration with the Experimental Cardiology department of the University of Amsterdam has demonstrated that this can be the case in Brugada syndrome, a syndrome that is responsible for many sudden cardiac deaths in young, apparently healthy subjects.
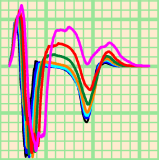
understanding electrograms
Our bidomain model of the human heart is the first model ever that can reliably simulate endocardial catheter signals. A first demonstration of this capability is our study of methods to measure repolarization times in cardiac electrograms [1,2].
We have used the model also to prove the validity of an extremely simple model of cardiac electrograms [3]. This simple model can help to gain insight in these signals.
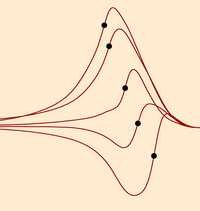
heterogeneity and the normal ECG
The shape of the ECG has been studied for more than a century, and explanations for most of its features have been given. However, when you actually try to reproduce an ECG with a mathematical model, it becomes clear how little we know about the actual mechanisms. This is a major problem for model developers, and at the same time a major reason for them to do their work: only when we point out the gaps in our knowledge we can pose the right questions and hope to improve our picture of the heart.
Especially the T-wave in the ECG is poorly understood. It depends crucially on heterogeneity of action potential duration in the heart. Several different cell types have been identified, but many of their characteristics are still debated. Moreover, when we put them together in the heart, we find that the information from measurements on single cells does not explain the shape of the T-wave; some unsupported assumptions have to be made to obtain a realistic T-wave. In addition, it turns out that we know too little about the placement of the different cell types in the heart.
| [1] | Trudel et al. IEEE Trans Biomed Eng 2004 |
| [2] | Potse et al, 34th Int Con Electrocardiol, June 2007 |
| [3] | Potse et al., Canadian Cardiovascular Congress. October 2007 |
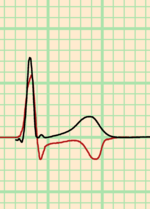
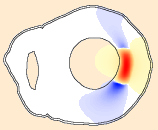
myocardial ischemia
"Myocardial ischemia" is the name of the complex changes that the heart muscle undergoes when it receives too little oxygen. This can be due to obstruction of a coronary artery. However, in many patients ischemic changes occur without a clear obstruction. These patients often have a particular ECG pattern, characterized by a negative shift of the "ST segment" of the ECG. In contrast, patients with a clear obstruction usually have a positive shift in their ST segments.
In the past, this negative ST-segment shift ("ST depression") has been attributed to ischemia limited to the inner part of the heart muscle. Recent computer model studies have indicated that such "subendocardial ischemia" does not lead to ST depression. Our study with a whole-heart model has confirmed this result [1]. We have also provided two alternative explanations: changes in the distribution of gap junctions, or an ischemic area that covers a very large part of the heart. For example, we have shown that ischemia covering the entire ventricular subendocardium can cause the typical ST depression pattern [2,3].
Another aspect of ischemia that we have studied is the distribution of potassium ions in the border of an ischemic area. Potassium plays an important role in the generation of ischemia-related ECG changes. We have shown that more realistic results can be obtained if the model accounts for the dynamics of potassium [4,5]. We are working on a reaction-diffusion model that completely integrates potassium handling. With this model we hope to improve understanding of cardiac arrhythmias that are caused by ischemia.